|
10/22/2023 0 Comments Iso 13485 design control 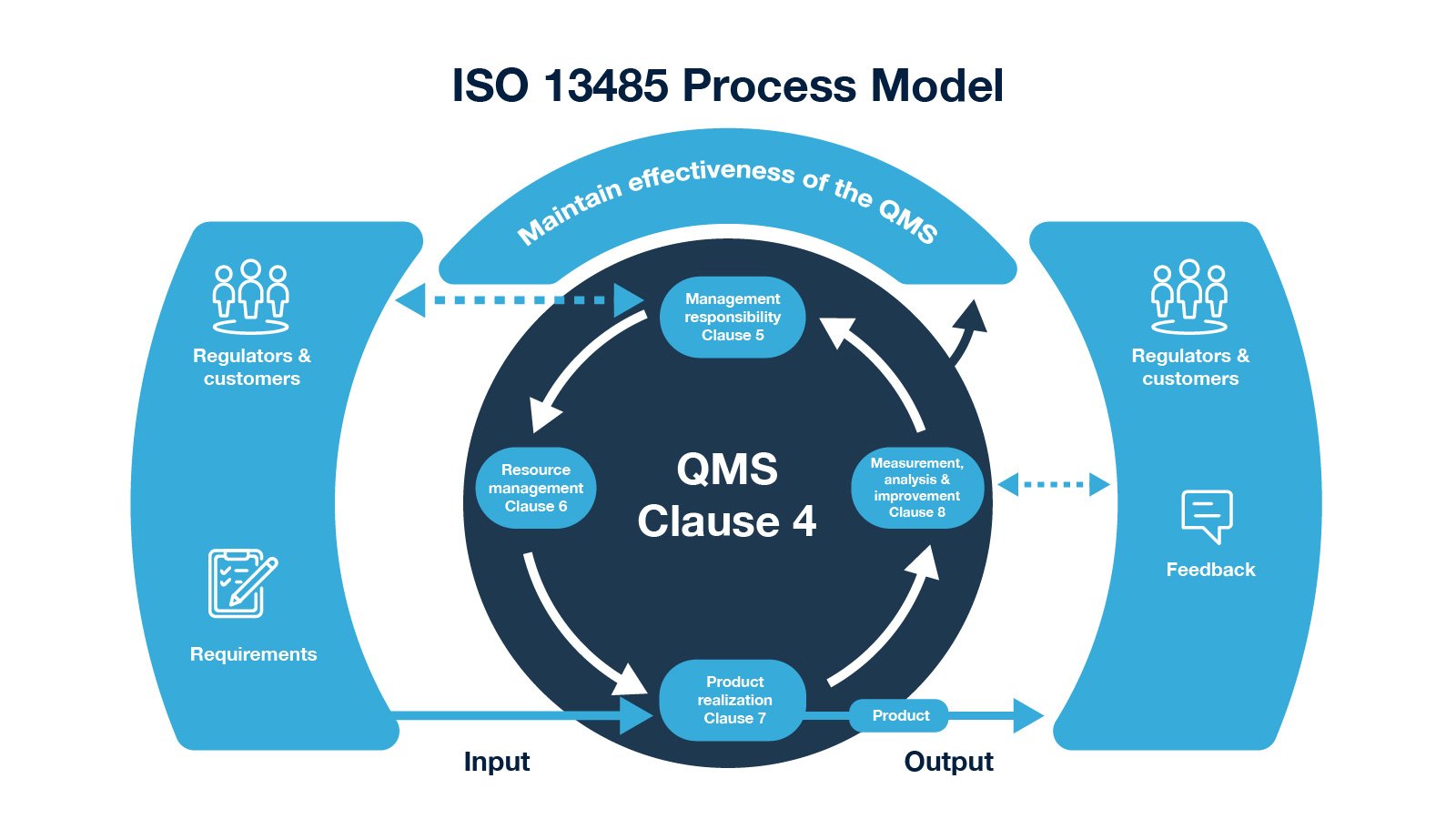 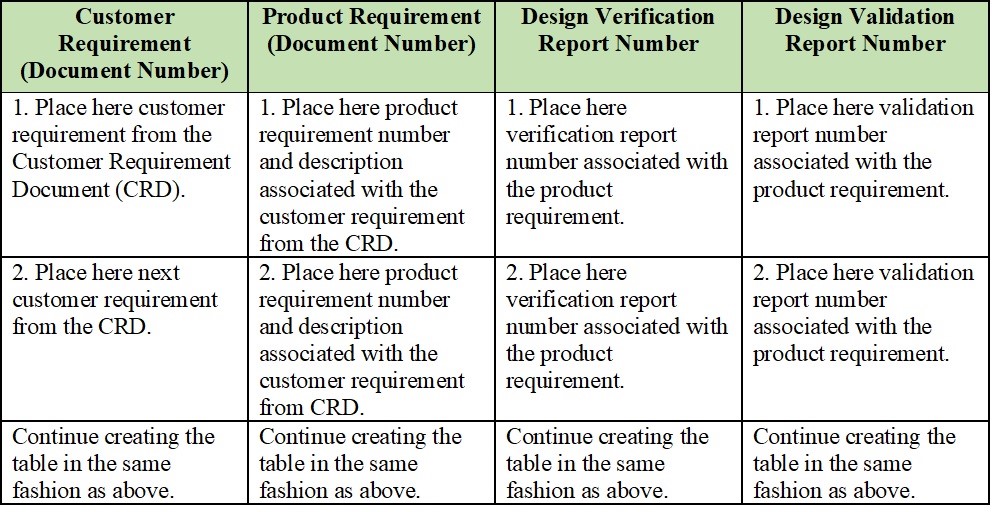
The point is that if you as a medical device company fail to meet regulatory design requirements, it won’t be able to reach the market.īecause Design and development processes include planning, inputs, outputs, review, verification, validation, transfer, control of D&D changes, and design and development files. 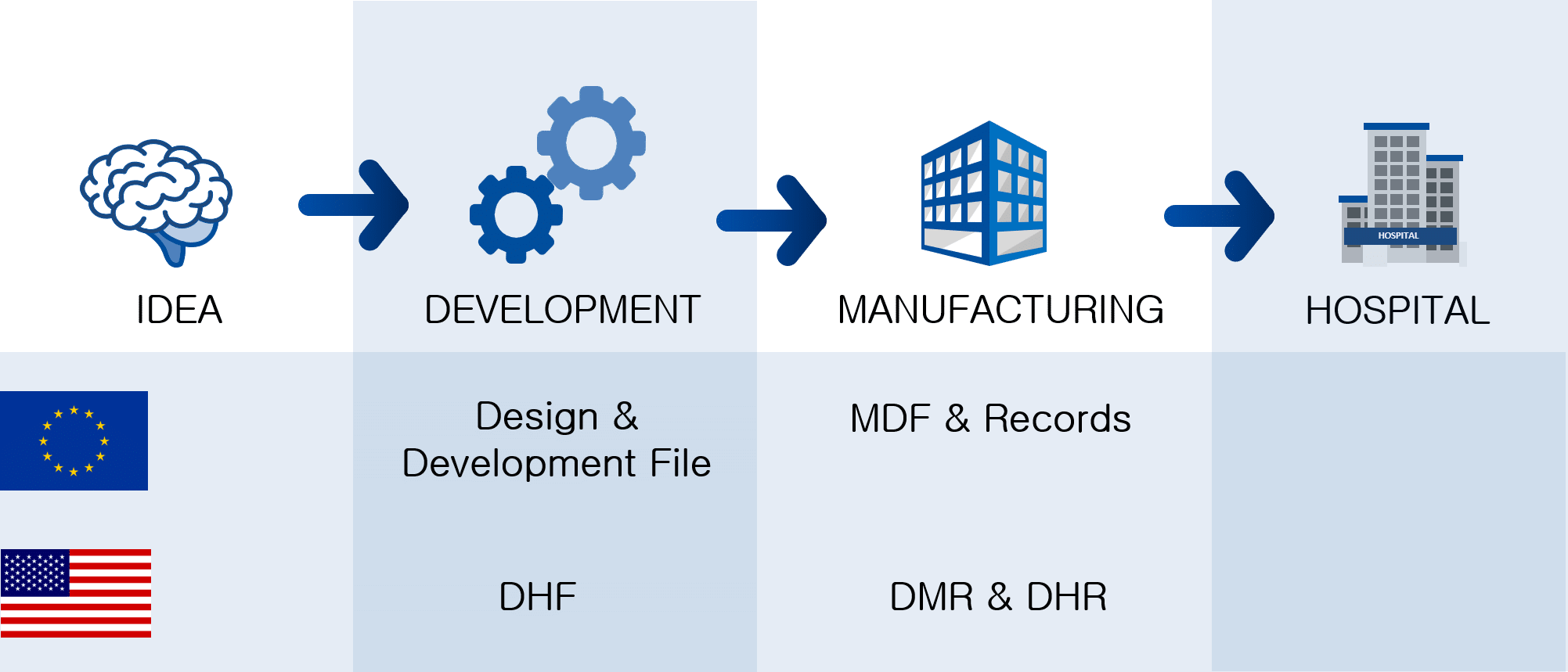
On one hand, it’s a continuation of the previous lesson based on customer needs and requirements that you included in the D&D process. The design and development process for medical devices has to deal with product safety, risk controls, relevant regulations, and customer needs. The design and development of the medical device are one of the most crucial parts of the ISO 13485 standard due to its complexity compared to other industries.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
